Artemisia annua is the dry aboveground part of Artemisia annua L., a compositae plant, it has a long history of medicinal use in China since more than 2,000 years ago. In 1971, Tu Youyou and other scientists discovered that artemisia annua extract had significant therapeutic effects on rat and monkey malaria, and named its effective monomer artemisinin, which was a milestone in the development of antimalarial drugs. Artemisinin is a sesquiterpene lactone compound with a peroxide bridge structure, which does not contain azacyclic structure. Compared with traditional antimalarial drugs such as chloroquine and quinine, artemisinin has better efficacy, lower toxicity and fewer adverse reactions. Its disadvantage is the high recurrence rate, but it can be solved by combining with other antimalarial drugs.
Artemisinin And Its Derivatives
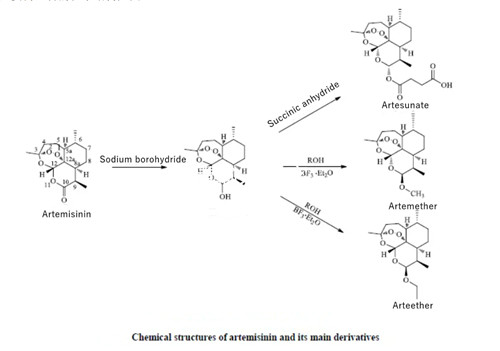
In order to improve the physicochemical properties of artemisinin, scientists studied its structure and synthesized dihydroartemisinin, artesunate, artemether, artemether and other derivatives. Artemisinin and its derivatives are sesquiterpene lactones. Deoxy artemisinin showed negative effects on P. bergeri mice, indicating that the biological activity of artemisinin was related to the structure of the peroxide bridge. Therefore, the structural modification of artemisinin is carried out based on preserving the peroxide bridge, mainly by modifying the structure of C-9 and C-10, with c-10 being the most common modification. This paper focuses on this artemisinin and its derivatives for your reference. The chemical structure of artemisinin and its main derivatives is shown below:
Artemisinin
Artemisinin is a colorless crystalline powder with the molecular formula C15H22O5. High resolution mass spectrometry analysis showed that the compounds for half terpene, infrared spectrum and its quantitative reaction with triphenylphosphine show that the compound existing in the special peroxide groups, nuclear magnetic resonance (NMR) and X-ray diffraction (XRD) technique to determine the structure of artemisinin and its relative configuration, obtained the lactone ring absolute configuration through the optical rotatory dispersion technology.
With the development of research, the pharmacological effects of artemisinin and its derivatives are not only limited to anti-malaria, but also found to be effective in anti-tumor, anti-inflammation, anti-fungal, anti-fibrosis and other aspects. In clinical application, artemisinin is found to have poor water solubility and lipid solubility, poor stability, low oral bioavailability, and only 3-5H plasma half-life, which limit its clinical application.
Dihydroartemisinin

Artesunate
Artesunate(C19O8H28) is synthesized by esterification of dihydroartemisinin and succinic anhydride. It has antimalarial, antiviral, anti-inflammatory, anti-tumor and immunomodulatory effects. Artesunate is a weakly acidic drug with high efficiency, low toxicity and resistance resistance. It is easily diffused in the body and permeates biofilms easily. With a pKa value of 3.5 ~ 5.5, artesunate is poorly ionized in acidic body fluids, but soluble in weakly alkaline solutions. Based on this characteristic, artesunate can be prepared as an injection, tablet, suppositories or other dosage forms for injection, oral or rectal administration.
Artemether and Arteether
Using dihydroartemisinin as substrate and replacing hydrogen atoms on c-10 hydroxyl groups with hydrocarbon groups, artemisinin ether derivatives were obtained, the most typical are artemether and artemether, both of which have higher activity than artemisinin. Artemisinin ether derivatives have good lipid solubility, but poor water solubility and low bioavailability. Direct injection is easy to cause irritation. Therefore, some scholars encapsulated artemisinin ether liposomes modified by aminopterin into targeted nano-liposomes modified by aminopterin, which are round and uniform and relatively stable. The results of in vitro release showed that the liposome could release drugs in vivo for a long time, and could improve the metabolism and bioutilization of artemether.
Other derivatives
Some structural analogues, polymers and structural simplifications of artemisinin have also been synthesized on the basis of the unique structure of artemisinin. Artemisinin dimers are composed of two artemisinin monomers linked by ligons. Commonly used ligons include alkyl, ether bond, ester group and amine carbonate, etc. Compared with artemisinin monomers, the dimers have strong pharmacological activity, small adverse reactions and good physical and chemical properties. Studies have shown that it has excellent anticancer activity in vitro and in vivo, and different ligands have great influence on its anticancer activity. Therefore, the properties of artemisinin dimers can be optimized by changing the ligand. Sodium artesunate is a basic salt with good water solubility, rapid action, good tolerance and can be used for intravenous or intramuscular administration.
Production and Synthesis of Artemisinin
Biosynthesis of artemisinin
Artemisinin is found in the flowers and leaves of artemisia annua, a compositae plant. According to current studies, the main synthetic pathways of artemisinin are meth-ylerythritol phosphate (MEP) and mevalonic acid (MEvalonic acid). The MVA pathway provides dimethylallyl diphosphate (DMAPP) and isopentenyl phosphate (IPP), Farnesyl diphosphate synthase (FPS) was then formed from one DMAPP and two IPPs. Amorpha 4,11-diene synthase (ADS) can be used as a catalyst to generate FPP. Amorpha is catalyzed by cytochromeP450 monooxygenase (CYP71AV1). Artemisinin, artemisinin aldehyde and artemisinic acid were synthesized by three steps of oxidation, respectively. The mechanism of artemisinin formation from artemisinic acid is not completely clear, and there are two main viewpoints at present: Artemisinic aldehyde delta-11(13) reductase (DBR2) is responsible for the formation of dihydroartemisinic aldehyde, which reacts with aldehydehydrogenase 1(ALdehydrogenase 1). ALDH1) catalyzed the formation of Dihydroartemisinic acid (DHAA). DHAA is the direct precursor of artemisinic acid, and finally generates artemisinic acid under the action of photooxidation. Second, artemisinin B is photooxidized from artemisinic acid, followed by dihydroartemisinin B, and finally artemisinin is formed.
Chemical synthesis of artemisinin
In 1983, Swiss scientists Schmid and Hofheinz completed the total chemical synthesis of artemisinin using (-) -isoomenthol as raw material through a 10-step reaction. However, due to the complex chemical structure of artemisinin, the production process of this method is complicated, the cost is high, the yield is low, and it is far from meeting the requirements of industrial production. Therefore, people focus on artemisinin acid, artemisinin B, artemisinin and other precursors for artemisinin semi-synthesis research.
Extraction and isolation of artemisinin
Due to the total synthesis and semi-synthesis of artemisinin, extraction, isolation and purification are still the main methods to obtain artemisinin in industry. Artemisinin is poor in water solubility, almost insoluble in water, soluble in ethanol, ether, easily soluble in chloroform, acetone, ethyl acetate and benzene and other organic solvents. The initial extraction methods were mainly organic solvent extraction followed by recrystallization or column chromatography separation and refining. In recent years, the extraction of artemisinin has been mainly adopted by supercritical fluid extraction, bio-complex enzyme extraction, ultrasound-assisted extraction and other new extraction methods, which have the advantages of high extraction rate, short heating time and low cost. Negi et al. used supercritical carbon dioxide to extract artemisinin from artemisia annua leaves, and measured the global yield isotherms of artemisinin. The highest yield was 3.65%. The desorption of supercritical carbon dioxide from the organic fraction of n-hexane can obtain a higher mass fraction of artemisinin.
Ultrasound-assisted extraction of artemisinin has been very common. A new organic solvent, hydrophilic deep eutectic solvent methyltrioctyl Ammonium chloride 1-butanol (N81Cl-NBA), has been used for ultrasound-assisted extraction of artemisinin. The extraction rate of artemisinin was about 8 mg/g, which was significantly higher than that of conventional organic solvent. The target artemisinin was recovered from n81Cl-NBA extract with AB-8 macroporous resin, and the recovery was 85.65%. The solvent is considered as a safe solvent for the extraction of bioactive substances and can be used in the pharmaceutical field.
Research: Synthetic biology of artemisinin

Artemisia Annua dried leaf
Synthetic biology integrates chemical engineering, biotechnology, medicine and other disciplines to optimize the synthesis pathway of natural products through the reconstruction, design and transformation of microbial chassis cells, and realize the initial synthesis of natural products. Artemisinin biosynthesis has become a hot topic.
Ro et al. modified S. cerevisiae and realized the synthesis of artemisinic acid, the late precursor of artemisinin, in S. cerevisiae with a mass concentration of 100 mg/mL. The synthesized artemisinic acid is excreted by engineered yeast, simplifying the purification process. Paddon et al. found that production of high levels of artemisinic acid also requires the involvement of cytochrome B5 (CYB5), ADH1 and ALDH1. By optimizing the expression ratio of CYP71AV1 and its homologous reductase cytochrome P450 reductase, combined with the expression of CYB5, the yield of artemisinic acid was 25g/L under the condition of co-expression of artemisinol and acetaldehyde dehydrogenase. At the same time, the group converted artemisinic acid into artemisinin using the chemical source of singlet oxygen, thus eliminating the need for specialized photochemical equipment. Meadows et al. used four non-natural metabolic reactions to alter the metabolism of central carbon in s. cerevisiae, and obtained β -farnene at least 100g/L in 6 days of fed-batch fermentation. This production method was 25% higher than that of the control strain, while requiring 75% less oxygen, which helped to reduce production costs. AaMYB15 is a r2R3-MyB transferrin that negatively regulates artemisinin biosynthesis. It directly binds to the promoter of AaORA, a positive regulator of artemisinin biosynthesis in the jasmonic acid signaling pathway, and inhibits its transcriptional activity, thereby reducing the expression level of downstream key enzyme genes and negatively regulating artemisinin biosynthesis. This discovery provides new insights into the regulation of artemisinin biosynthesis.
- Dandelion Extract: What It Is, Benefits, Uses and Side Effect - April 23, 2024
- Is Berberine Extract Help For Weight Loss? - April 11, 2024
- Why Is Pysllium Husk Powder A Popular Meal Replacement Ingredient? - April 3, 2024